The hemp plant makes more than 100 distinct molecules that researchers classify as cannabinoids. That number, large as it sounds, is less interesting than the fact that almost all of them descend from a single parent compound. Understanding cannabinoids is easier when you think of the family as a tree with a common root, rather than a random cabinet of acronyms.
This essay walks through the cannabinoid family at the level a curious reader actually needs: where these compounds come from in the plant, how they change when exposed to heat or time, and what the established research says about the major members. It is not a medical reference. It is closer to a field guide.
The parent molecule
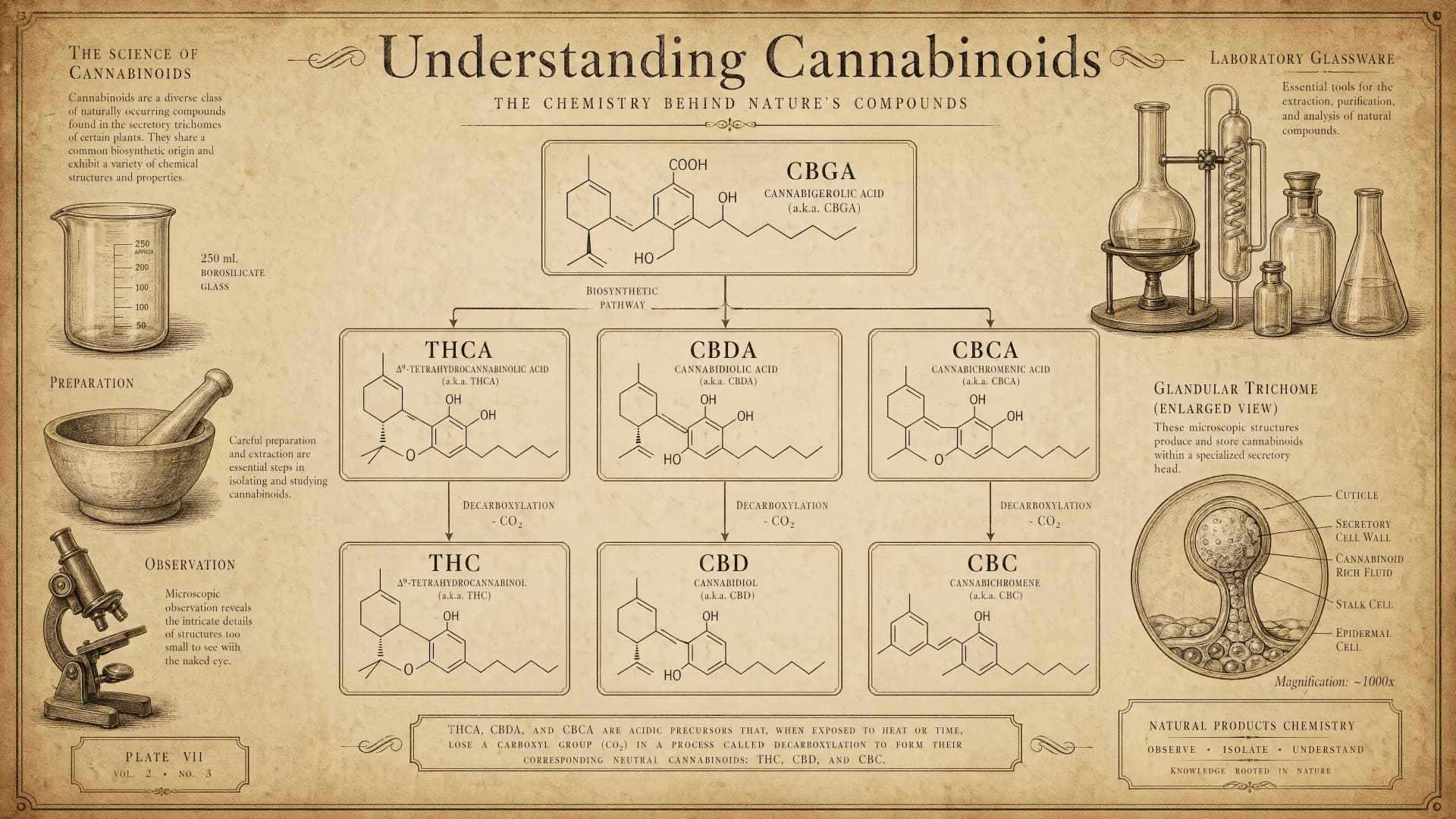
Inside the plant, cannabinoid biosynthesis starts with a compound called cannabigerolic acid, or CBGA. CBGA is sometimes nicknamed the "mother" cannabinoid because downstream enzymes convert it into the acidic forms of most other cannabinoids: THCA, CBDA, CBCA, and so on. The letter "A" at the end of each acronym stands for "acid," and it matters. Acidic cannabinoids behave differently in the body than their neutral counterparts.
If you pick a fresh hemp flower straight from the plant and analyze it, the vast majority of the cannabinoid content will be in acid form. The neutral cannabinoids that dominate popular discussion — plain CBD, plain CBG, plain THC — are largely created by decarboxylation, a heat-driven reaction that removes a carboxylic acid group from the molecule.
What decarboxylation does
Decarboxylation is the single most important chemical event in the journey from raw plant to consumable product. When an acidic cannabinoid is exposed to sustained heat — a flame, an oven, a vaporizer — the acid group is lost as carbon dioxide and the molecule converts to its neutral form. CBDA becomes CBD. CBGA becomes CBG. THCA becomes THC.
This is why a raw tincture made from fresh plant matter can have a very different pharmacological profile from a dried, heated flower sample of the same source material, even when the total cannabinoid content is identical on paper.
The major cannabinoids
CBD (cannabidiol)
CBD is the most extensively studied cannabinoid outside of THC. Its binding affinity at the CB1 receptor is weak, which is part of why it does not produce the intoxication associated with THC. Its known activity includes allosteric modulation of the CB1 receptor (reducing THC's effects when the two are co-administered), direct action at 5-HT1A serotonin receptors, and TRPV1 channel activity. The FDA has approved one CBD-based pharmaceutical, Epidiolex, for specific pediatric seizure disorders; the evidence for everything else CBD is popularly used for ranges from suggestive to preliminary.
CBG (cannabigerol)
CBG is present in most commercial samples at low concentrations — often under 1% — because most of the CBGA in the plant gets converted to downstream cannabinoids before harvest. CBG-dominant chemotypes are a recent breeding achievement; they came about by selecting for genetics that knock out the enzymes converting CBGA to other acidic cannabinoids. Research on CBG is early but active; it is a partial agonist at CB1 and CB2 receptors and shows activity at alpha-2 adrenergic and 5-HT1A receptors in vitro.
CBN (cannabinol)
CBN is an oxidation product, not a biosynthesized cannabinoid. When THC is exposed to oxygen and time, it slowly degrades to CBN. Older, less-carefully-stored flower tends to test higher in CBN for this reason. CBN has some binding affinity at CB1 receptors but much lower than THC. The popular "sedative" reputation of CBN is not well supported by controlled human research; what does seem to be true is that samples high in CBN are usually also samples where the overall terpene profile has degraded, which may explain some of the reported sedation.
CBC (cannabichromene)
CBC is present in small amounts in most chemotypes. It has very weak CB1 affinity but shows interesting activity at TRP (transient receptor potential) channels, which are involved in sensation, including pain and temperature perception. CBC is one of the quieter members of the family, under-researched relative to its likely relevance.
Acidic cannabinoids
CBDA, CBGA, and their relatives are increasingly interesting to researchers. They have different binding profiles than their decarboxylated counterparts and, in some cases, higher potency at specific receptors. CBDA, for example, is a much stronger 5-HT1A agonist than CBD. This is one reason "raw" or cold-pressed preparations have attracted some research attention.
Receptors and the endocannabinoid system
The two primary receptors that cannabinoids interact with in humans are CB1 and CB2. CB1 is dense in the central nervous system, especially in areas controlling coordination, memory, and reward. CB2 is denser in peripheral tissues, especially immune cells. The endogenous ligands for these receptors — compounds your body makes on its own — include anandamide and 2-AG. These compounds form what researchers call the endocannabinoid system.
The discovery that the body has its own cannabinoid-signaling machinery is relatively recent (1990s) and still reshaping how researchers think about appetite, mood, pain, and memory. Cannabinoids from plants interact with this pre-existing system; they are not foreign to it.
What the research doesn't yet settle
Three areas to read with a skeptical eye.
Dosing. Because cannabinoid bioavailability varies dramatically by route of administration (inhaled vs. oral vs. sublingual) and by the lipid content of the delivery vehicle, dose-response curves that look clean in one study don't translate cleanly to another. When a study reports "25 mg CBD," the amount that actually reaches circulation can range from under 5% to over 30%.
Interactions. Cannabinoids interact with the cytochrome P450 enzyme system, which metabolizes many common medications. CBD in particular can inhibit CYP3A4 and CYP2C19. This is not a reason to panic, but it is a reason to discuss cannabinoid use with a physician if you are on other medications.
Entourage claims. The hypothesis that whole-plant preparations outperform single-cannabinoid preparations is plausible and widely believed, but the controlled evidence is still thinner than the enthusiasm. Two studies can produce opposite results depending on the ratios they test.
How we read a cannabinoid panel
A competent lab report should show the concentration of each cannabinoid in both acid and neutral form, plus a total for each family. When we look at a panel, we pay attention to:
- The ratio of acidic to neutral forms, which tells us how the sample has been handled or cured.
- The presence of minor cannabinoids (CBG, CBC, CBN) rather than just the headline values.
- Whether the lab reports the limit of detection. Any value below LOD should be treated as "not detected," not as zero.
We talk more about reading lab reports in our essay on lab testing and transparency.